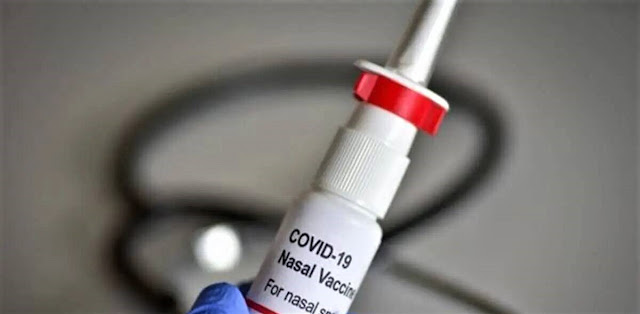
Representational image. Photo: Collected
The world's first intra-nasal Covid vaccine developed by India has got approval from the country's Central Drugs Standard Control Organisation for restricted use.
The vaccine will be used as booster doses for people aged 18 & above only in emergency situations.
The vaccine, which successfully passed clinical trials, has been developed by Bharat Biotech International Limited (BBIL), reports our New Delhi correspondent.
The intra-nasal vaccine was evaluated in Phases I, II & III clinical trials with successful results, the Indian health ministry said in a statement on Thursday, adding the phase-III trials of the intra-nasal vaccine were conducted on 3,100 people for safety & immunogenicity.
The nasal delivery system has been designed & developed to be cost-effective in low- & middle-income countries. It is stable at 2-8°C temperature for easy storage & distribution, the ministry statement said.
Comments
Post a Comment